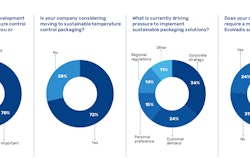
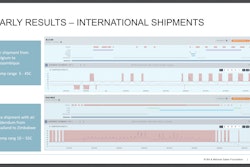
This content was written and submitted by the supplier. It has only been modified to comply with this publication’s space and style.
Aphena Pharma Solutions announces that it now offers cold chain storage and large-scale biologics packaging and distribution in a new, 500,000-square-foot property that is purpose-built for FDA manufacturing and packaging operations. This new facility is part of the company’s recently announced $21 million expansion of its Solid Dose Division in Tennessee.
Within that new space, Aphena has dedicated 47,000 square feet to cold chain storage, with 14,000 pallet spaces available. This space features 24-hour, state-of-the-art security; redundant power supplies with a failover compressor; temperature mapping and monitoring; automatic alarm notification; and chilled storage and distribution with temperatures ranging from 2º C to 8º C refrigerated and -10º C to -18º C frozen.
“This addition of cold chain storage, turnkey biologics packaging and distribution services establishes Aphena’s position as a top contender in the pharmaceutical contract service space,” said Eric Allen, Aphena’s EVP of Sales. “These new service offerings, on top of our recently expanded solids and liquids operations, will continue to bring added value to future and current customers worldwide.”
An additional benefit of Aphena locating its cold chain storage and biologics operations in Tennessee, the distribution hub of the U.S., is that it is less than 500 miles away from 75% of the U.S. population.
“Aphena is very excited to announce this new level of offerings to better serve our customers with expanded project capabilities at every level,” said Aphena CEO Shawn Reilley. “This addition effectively makes Aphena the largest provider of cold chain storage and one of the most diversified manufacturing and packaging companies in the pharmaceutical industry.”
With two separate FDA- and DEA-registered locations in the U.S., Aphena handles solid dose, liquids, gels, creams, ointments, foams, suspensions and lotion-based products.