Healthcare Distribution Alliance’s (HDA) 2018 Traceability Seminar plans to provide updates to its Verification Router Service (VRS) during an interactive panel discussion.
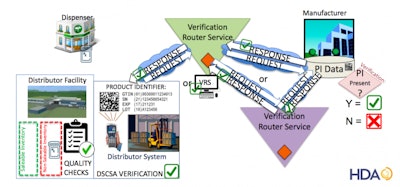
Since 2016, HDA has convened industry participants to develop the technical specifications necessary to build a VRS network. With nearly 60 million saleable returns processed annually, the existence of the VRS network will provide a critical method for the industry to comply with the 2019 saleable returns verification requirement.
During the session, VRS testing participants will discuss what the network is, share what they have learned through the testing process to date, what to consider as companies set up their system to connect to the VRS network, how and when to connect, and critical success factors. Attendees will have the opportunity to share their experiences and provide feedback through facilitated discussions and polling.
Session panelists will include:
- David Colombo, Director, Life Sciences Advisory Services, KPMG, LLP;
- Kevan R. MacKenzie, Director, Serialization Technology, McKesson Corp.;
- Matt Sample, Senior Director, Secure Supply Chain, AmerisourceBergen Corp.; and,
- Allison Sheldon, Senior Manager, Business Technology, Pfizer Inc.
The Oct. 17-19 event will take place at the Renaissance Washington, DC Downtown hotel. Full session descriptions are available here.
2018 seminar sponsors include Accenture, Adents, Antares Vision North America, LLC, Arvato Systems N.A., Axway, BlueLink Associates, Cardinal Health Specialty Solutions, DMLogic LLC, Enterprise System Partners Global Corporation, Five Rivers RX, HDA Origin, KNAPP Inc., LSPediA, Mettler-Toledo PCE Track and Trace, rfxcel, TraceLink Inc., TrackTraceRx, TruTag Technologies, Inc., VAI (Vormittag Associates, Inc) and Vantage Consulting Group.
For more information, contact:
Registration: HDA’s Meetings & Conferences Department, (703) 885-0278.
Programming: Subrina Ghorashi, (703) 885-0273.
Sponsorship and Table-Tops: Lisa Kanfer, (703) 885-0270.