At the 2013 OSIsoft Users Conference in San Francisco recently, consultant Dennis Brandl gave a talk to pharmaceutical manufacturers about “enterprise recipe management” (ERM), a method for using the ISA-88 and IEC 61512-3 standards for recipe management and the MESA BatchML V6.0 standards to improve communication and manage product definitions across internal and extended supply chains.
Brandl is a former chairman of the ISA SP88 Batch System control standard, a member of ISA’s SP95 Enterprise/Control System Integration committee, and a USA expert on batch control to the International Electrotechnical Commission (IEC). He says ERM is a strategy to support faster technology transfers, better product data collection and faster investigations, and is a way to reduce the time and effort required to build manual and electronic batch records.
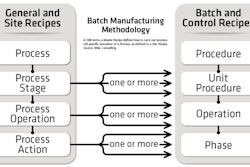
ERM takes ISA-88 recipe standards and extends them to the enterprise via ISA-95 material, equipment, and personnel standards. “It works because, for any type of production, there are about 50 to 70 basic actions that define a company’s basic production capability. Everything that’s done in the laboratory and in production facilities can be defined using these basic actions,” Brandl says. “About half of all actions are common across industries, and half are actions unique to production types.”
Implementing an ERM strategy can improve products and speed up delivery, Brandl said. It can also improve conformance with regulatory compliance mandates by standardizing the information for compliance investigations.
“Process reports can define information that must be collected and reported on during execution of a process—regardless of the equipment layout or level of automation,” he says. “Formalizing the minimum amount of information that must be collected and made available significantly reduces the time and effort required to perform investigations.” Process reports also can be reused for the same processes at different sites, enhancing enterprise-wide compliance.
The ANSI/ISA-88 standard (also known as IEC 61512-3 or S88, for short) enables a universal model for batch control that eliminates the difficulty of communicating requirements and integrating systems from different vendors. As such, it is facilitating the development of new “recipe-based” systems in many industries.
The MESA BatchML V6.0 standard defines a common XML format for exchanging information about recipes, the S88 equipment model, and batch production records. BatchML is a conforming XML implementation of the ISA-88 data standards. It has been used by many companies to archive their control system information in a vendor-independent format.
“Because it is XML, the data is easily viewable in a normal browser, or can be easily formatted for reports, without having to have access to the original system. This simplifies process analysis and investigations when the underlying control systems change,” says Brandl. “Some vendors are also using the Batch ML format as their internal archive and restore format, simplifying their integration to other systems and making software updates easier.”