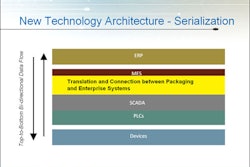
In a July guidance document to industry, the FDA set forth some suggestions for incorporating so called “physical-chemical identifiers” (PCIDs) into solid oral dosage-form drug products and paid particular attention to those that might migrate from packaging or container labeling.
The FDA defines a PCID as a product with some unique physical and or chemical characteristic that is added to the dosage form of a drug product and which “makes it possible to detect and authenticate legitimate dosage forms and identify counterfeits.”
These so-called PCIDs can include inks, pigments, flavors, or molecular taggants, says the FDA. And the agency expects that many of them would be made of existing substances used in food additives, colorants, or drug products excipients. The FDA's guidance document, entitled “Incorporation of Physical-Chemical Identifiers Into Solid Oral Dosage Form Drug Products For Anticounterfeiting,” is primarily concerned with helping drug makers and applicants for new drug approvals incorporate relevant information about their PCIDs into new drug applications, supplemental applications, and annual reports submitted to the FDA.
The guidance does not attempt to address radio frequency identification (RFID).
The FDA has been addressing the problem of counterfeit drugs since at least 2004, when it produced its “Combatting Counterfeit Drugs” report. Counterfeits have tended to be a bigger problem overseas than in the U.S., though it is nevertheless a concern here, and not just when drugs are imported. The FDA has always recognized the importance of packaging technologies as potential solutions, including but not limited to RFID.
Confirming the authenticity of any individual package of drugs, or, when possible, even an individual dosage, is the essence of thwarting counterfeits. In the complex distribution network through which drug products travel, opportunities abound to introduce counterfeit product into the mix. Counterfeits can come in many forms. For example, they might be products that are just imitations, and literally lack the right ingredients, or are out-of-date versions of the real thing, or are otherwise diverted from the ordinary authorized distribution pathways. It is a recognized phenomenon that authentic and appropriate products are often mixed with inauthentic counterfeits, meaning the confirmation of authenticity often involves measures going to the individual dosage level. Hence, ideas like PCIDs.
As for PCIDs that are added to packaging or labeling of drug products in solid oral dosage form, the FDA makes specific recommendations on how to evaluate toxicological concerns and offers guidance on when and how to report or request approval of the addition of a PCID. The agency points out that, as expected, its “toxicological concerns are mitigated if the added substance(s) is a permitted direct or indirect food additive,” or listed in the FDA's Inactive Ingredient Guide.
If the substance you want to use as a PCID in packaging is not the subject of known toxicological information, the packager should, says the FDA, determine that there won't be migration of the substance into the products. If some interaction of the PCID with the product is expected, and toxicological concerns may result, the FDA suggests packagers contact the agency to discuss the issues. If the toxicology of the substance hasn't been established, and the potential for its migration exists, the FDA says the drug product with that PCID in it would require a prior approval supplement before marketing.
When it comes to PCIDs in solid oral dosage form products, the FDA is hoping to help industry follow one of its key pieces of advice: Use a multifaceted approach to counterfeiting, in order to stay at least one step ahead of counterfeiters.
-By Eric F. Greenberg, Attorney-at-Law. Published in Packaging World magazine, Oct. 2009, p. 22. Eric can be reached at [email protected], or at his firm's Web site at www.ericfgreenbergpc.com.
The FDA defines a PCID as a product with some unique physical and or chemical characteristic that is added to the dosage form of a drug product and which “makes it possible to detect and authenticate legitimate dosage forms and identify counterfeits.”
These so-called PCIDs can include inks, pigments, flavors, or molecular taggants, says the FDA. And the agency expects that many of them would be made of existing substances used in food additives, colorants, or drug products excipients. The FDA's guidance document, entitled “Incorporation of Physical-Chemical Identifiers Into Solid Oral Dosage Form Drug Products For Anticounterfeiting,” is primarily concerned with helping drug makers and applicants for new drug approvals incorporate relevant information about their PCIDs into new drug applications, supplemental applications, and annual reports submitted to the FDA.
The guidance does not attempt to address radio frequency identification (RFID).
The FDA has been addressing the problem of counterfeit drugs since at least 2004, when it produced its “Combatting Counterfeit Drugs” report. Counterfeits have tended to be a bigger problem overseas than in the U.S., though it is nevertheless a concern here, and not just when drugs are imported. The FDA has always recognized the importance of packaging technologies as potential solutions, including but not limited to RFID.
Confirming the authenticity of any individual package of drugs, or, when possible, even an individual dosage, is the essence of thwarting counterfeits. In the complex distribution network through which drug products travel, opportunities abound to introduce counterfeit product into the mix. Counterfeits can come in many forms. For example, they might be products that are just imitations, and literally lack the right ingredients, or are out-of-date versions of the real thing, or are otherwise diverted from the ordinary authorized distribution pathways. It is a recognized phenomenon that authentic and appropriate products are often mixed with inauthentic counterfeits, meaning the confirmation of authenticity often involves measures going to the individual dosage level. Hence, ideas like PCIDs.
As for PCIDs that are added to packaging or labeling of drug products in solid oral dosage form, the FDA makes specific recommendations on how to evaluate toxicological concerns and offers guidance on when and how to report or request approval of the addition of a PCID. The agency points out that, as expected, its “toxicological concerns are mitigated if the added substance(s) is a permitted direct or indirect food additive,” or listed in the FDA's Inactive Ingredient Guide.
If the substance you want to use as a PCID in packaging is not the subject of known toxicological information, the packager should, says the FDA, determine that there won't be migration of the substance into the products. If some interaction of the PCID with the product is expected, and toxicological concerns may result, the FDA suggests packagers contact the agency to discuss the issues. If the toxicology of the substance hasn't been established, and the potential for its migration exists, the FDA says the drug product with that PCID in it would require a prior approval supplement before marketing.
When it comes to PCIDs in solid oral dosage form products, the FDA is hoping to help industry follow one of its key pieces of advice: Use a multifaceted approach to counterfeiting, in order to stay at least one step ahead of counterfeiters.
-By Eric F. Greenberg, Attorney-at-Law. Published in Packaging World magazine, Oct. 2009, p. 22. Eric can be reached at [email protected], or at his firm's Web site at www.ericfgreenbergpc.com.