This content was written and submitted by the supplier. It has only been modified to comply with this publication’s space and style.
Multisorb Technologies (Multisorb), the world's leading active packaging technology company, today announced at Interphex 2010 the introduction of SimulOxSM, a pseudo-empirical modeling service that helps to determine the components of sorbents used to manage the oxygen environment in pharmaceutical packaging. Multisorb is located at booth #644 at Interphex, April 20-22, at the Jacob K. Javits Convention Center in New York City.
“Many pharmaceutical manufacturers make a fundamental and costly mistake when introducing new drugs,” says Adrian Possumato, Global Director of Healthcare Packaging at Multisorb. “They spend millions developing fantastic medicines and then run into problems when they find they are unable to manage the oxygen environment of the packaged drug product and ultimately limiting its shelf life and stability. The launch of their new product is pushed out as they try to find a quick fix to their challenge. This costs them time, market position, brand presence and ultimately profitability.”
SimulOxSM is a component of Multisorb's Calculation through OperationsSM (CtO) value proposition. As a simulation service, it uses sophisticated mathematical formulations to analyze the drug product to be packaged and predict its chemical and/or physical stability outcomes when exposed to oxygen in the headspace volume of a container. It then identifies the elements needed to formulate a tailor-made sorbent with total accuracy, which will protect the integrity of the drug product concerned. It also determines if the solution should be a drop-in, fit-in, or a built-in intelligent sorbent.
“To obtain optimal results during the consultative SimulOxSM process, our scientists collaborate with the pharmaceutical company's formulation chemists, packaging and equipment engineers. Their objective is to determine the correct sorbent solution to enhance the product's stability, integrity and shelf life. Not only is this a good technical decision, it is also a great business strategy as any potential hiccups would be taken care of long before the product went into full production,” adds Possumato.
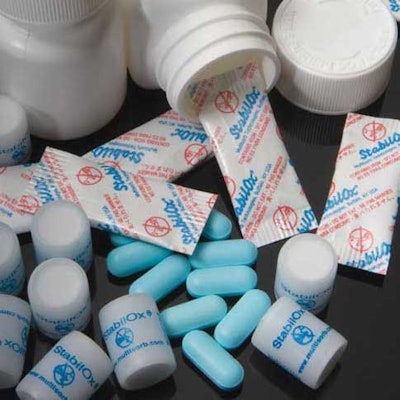
Based on the outcome of the SimulOxSM simulations, Multisorb specialists then formulate the correct sorbent solution, which is packaged as a product called StabilOx® and is delivered in oxygen absorbing packets, labels and canisters. These are ideal for pharmaceutical formulations that are susceptible to oxidative degradation and significantly improved stability in comparison to desiccant use to control moisture-mediated oxidation. They can also be used in medical device packaging.
By following this best practice process, pharmaceutical manufacturers can achieve true Quality by Design (QbD) standards and potentially get the new product to market six to 12 months sooner.
In addition, Multisorb has a pseudo-empirical modeling program and simulation service called SimulSorbSM, which is also used as part of the CtO process to determine the best approach in managing moisture levels in packaging. It is often used in conjunction with SimulOxSM.
Multisorb question-and-answer document
About SimulOxSM and StabilOx®
1. What is SimulOxSM?
SimulOxSM is a pseudo-empirical modeling service that helps determine the sorbents required to manage the oxygen environment in pharmaceutical packaging. This helps to reduce the rate of or otherwise eliminate oxidative degradation in drug substances, drug products, and medical devices.
2. How does the pseudo-empirical modeling service work?
SimulOxSM is a simulation service and a component of Multisorb's Calculations through OperationsSM (CtO) value proposition. It uses sophisticated mathematical formulations to analyze, for example, a drug product and its packaging. Based on this analysis, it can predict the drug's chemical stability outcome when exposed to oxygen in the headspace volume of a container. As a result, it is able to accurately identify the elements needed to formulate a tailor-made sorbent and determines if the solution should be a drop-in, fit-in, or a built-in intelligent sorbent to protect the integrity of the drug.
3. What is StabilOx®?
StabilOx® Specialty Oxygen Absorbers are literally the sorbent solution or product recommendation resulting from the SimulOxSM simulations. It is formulated by Multisorb specialists, and it can be delivered in oxygen absorbing packets, labels and canisters. These are ideal for pharmaceutical formulations that are susceptible to oxidative degradation and significantly improved stability in comparison to desiccant use to control moisture-mediated oxidation. They can also be used in medical device packaging.
4. How does it work?
StabilOx® does three things:
· It absorbs oxygen and effectively reduces package headspace oxygen levels
· It reduces or eliminates oxidative degradation
· It manages headspace relative humidity and drug product free moisture to reduce the rate of all chemical pathways of degradation
5. What is SimulSorbSM?
SimulSorbSM is also a pseudo-empirical modeling program and simulation service that is often used in conjunction with SimulOxSM as part of the CtO process to determine the best approach in managing moisture levels in packaging.
“Many pharmaceutical manufacturers make a fundamental and costly mistake when introducing new drugs,” says Adrian Possumato, Global Director of Healthcare Packaging at Multisorb. “They spend millions developing fantastic medicines and then run into problems when they find they are unable to manage the oxygen environment of the packaged drug product and ultimately limiting its shelf life and stability. The launch of their new product is pushed out as they try to find a quick fix to their challenge. This costs them time, market position, brand presence and ultimately profitability.”
SimulOxSM is a component of Multisorb's Calculation through OperationsSM (CtO) value proposition. As a simulation service, it uses sophisticated mathematical formulations to analyze the drug product to be packaged and predict its chemical and/or physical stability outcomes when exposed to oxygen in the headspace volume of a container. It then identifies the elements needed to formulate a tailor-made sorbent with total accuracy, which will protect the integrity of the drug product concerned. It also determines if the solution should be a drop-in, fit-in, or a built-in intelligent sorbent.
“To obtain optimal results during the consultative SimulOxSM process, our scientists collaborate with the pharmaceutical company's formulation chemists, packaging and equipment engineers. Their objective is to determine the correct sorbent solution to enhance the product's stability, integrity and shelf life. Not only is this a good technical decision, it is also a great business strategy as any potential hiccups would be taken care of long before the product went into full production,” adds Possumato.
Based on the outcome of the SimulOxSM simulations, Multisorb specialists then formulate the correct sorbent solution, which is packaged as a product called StabilOx® and is delivered in oxygen absorbing packets, labels and canisters. These are ideal for pharmaceutical formulations that are susceptible to oxidative degradation and significantly improved stability in comparison to desiccant use to control moisture-mediated oxidation. They can also be used in medical device packaging.
By following this best practice process, pharmaceutical manufacturers can achieve true Quality by Design (QbD) standards and potentially get the new product to market six to 12 months sooner.
In addition, Multisorb has a pseudo-empirical modeling program and simulation service called SimulSorbSM, which is also used as part of the CtO process to determine the best approach in managing moisture levels in packaging. It is often used in conjunction with SimulOxSM.
Multisorb question-and-answer document
About SimulOxSM and StabilOx®
1. What is SimulOxSM?
SimulOxSM is a pseudo-empirical modeling service that helps determine the sorbents required to manage the oxygen environment in pharmaceutical packaging. This helps to reduce the rate of or otherwise eliminate oxidative degradation in drug substances, drug products, and medical devices.
2. How does the pseudo-empirical modeling service work?
SimulOxSM is a simulation service and a component of Multisorb's Calculations through OperationsSM (CtO) value proposition. It uses sophisticated mathematical formulations to analyze, for example, a drug product and its packaging. Based on this analysis, it can predict the drug's chemical stability outcome when exposed to oxygen in the headspace volume of a container. As a result, it is able to accurately identify the elements needed to formulate a tailor-made sorbent and determines if the solution should be a drop-in, fit-in, or a built-in intelligent sorbent to protect the integrity of the drug.
3. What is StabilOx®?
StabilOx® Specialty Oxygen Absorbers are literally the sorbent solution or product recommendation resulting from the SimulOxSM simulations. It is formulated by Multisorb specialists, and it can be delivered in oxygen absorbing packets, labels and canisters. These are ideal for pharmaceutical formulations that are susceptible to oxidative degradation and significantly improved stability in comparison to desiccant use to control moisture-mediated oxidation. They can also be used in medical device packaging.
4. How does it work?
StabilOx® does three things:
· It absorbs oxygen and effectively reduces package headspace oxygen levels
· It reduces or eliminates oxidative degradation
· It manages headspace relative humidity and drug product free moisture to reduce the rate of all chemical pathways of degradation
5. What is SimulSorbSM?
SimulSorbSM is also a pseudo-empirical modeling program and simulation service that is often used in conjunction with SimulOxSM as part of the CtO process to determine the best approach in managing moisture levels in packaging.
Companies in this press-release





















