This content was written and submitted by the supplier. It has only been modified to comply with this publication’s space and style.
GS1 US has published a new guideline to assist the U.S. healthcare companies in using radio frequency identification (RFID) to improve supply chain visibility, efficiency, and consumer safety.
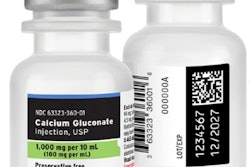
The “Implementation Guideline for RFID in Healthcare Manufacturing: Using GS1 Standards to Enable Visibility and Efficiency” document defines the application of GS1 Standards to support adoption of RFID by healthcare supply chain stakeholders. It informs pharmaceutical and medical device manufacturers about how to encode RAIN RFID tags using GS1’s Electronic Product Code (EPC) schemes outlined in the EPC Tag Data Standard (TDS) for automatic data capture to be utilized across the healthcare supply chain.
“Automation enabled by RFID not only offers inventory visibility, but also supports critical supply chain processes including withdrawals, product safety holds, return logistics, and more,” says Angela Fernandez, vice president of community engagement, GS1 US. “In these times of short supply of goods, knowing what is on hand and safe to use is a significant positive outcome for businesses and patients alike.”
The new guideline was developed by GS1 US healthcare workgroups to identify how suppliers should encode GS1 Standards in RAIN RFID tags and to provide a roadmap for adoption. The new document is designed to guide companies with implementing open, interoperable GS1 Standards to enable more efficient tracking, management, and traceability of products throughout the supply chain.
Distributors can learn how to integrate RFID technology within their systems to ensure compliance with new standards, and end users can benefit from understanding the available data and the access this enhanced method of data capture provides.