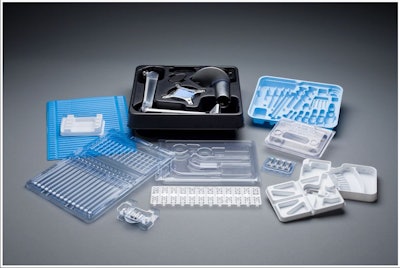
A recent Plastics Today article noted that Massachusetts-based UFP Technologies, maker of custom pieces for the medical, automotive, aerospace, and defense markets, has expanded their FlexShield product line for sterile barrier integrity protection for most medical devices. FlexShield is composed of flexible, medical grade thermoplastic polyurethane films that are both biodegradable and FDA approved.
FlexShield is also puncture and abrasion resistant, and can be formed into unique shapes, making it ideal for protecting medical devices and implants. The proprietary product line is approved for skin contact and can be used to guard patients from infection during surgery and safeguard sensitive medical equipment. Current applications include ergonomically designed pouches, tip protectors for sharp or delicate instruments, and bags that ensure sterile barrier integrity.