WPO (World Packaging Organisation) announced its 214 winners of the WorldStar 2024 global packaging competition. Winners were selected from 435 entries from 41 countries.
The Top Five winners, by country, are Japan (28), Germany (21), Australia and New Zealand (16), USA (12), and Brazil and Austria (11 each).
Says Luciana Pellegrino, WPO President, “This global award raises the voice for packaging and brings light to innovation, aligned to the main supply and consumption chains trends and modern life needs and challenges: save food, sustainability, branding and UX (user experience), food safety, smart packaging, among other aspects.”
Here are the Medical and Pharmaceutical category winners, in no particular order:
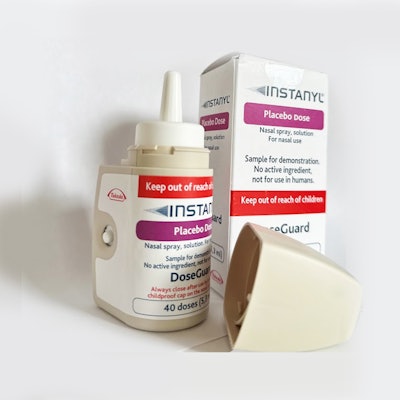
Instanyl DoseGuard Nasal Spray Device from Takeda Pharmaceuticals, Aptar Pharma (USA)
 World Packaging Organisation
World Packaging Organisation
The Instanyl DoseGuard is an electronic nasal spray lockout device developed to manage breakthrough pain in adults already using a controlled drug substance to control long-term cancer pain. “The aim of the Instanyl DoseGuard is to safeguard patients and others from the risk of overdose, misuse, abuse, addiction, diversion, accidental exposure, and medication errors,” reports the entry. “Instanyl DoseGuard is the first electronic nasal lockout device approved by a Notified Body and EMA for Takeda Pharmaceuticals International AG. It limits the number of doses a patient can take within the prescribed dosing regime and automatically locks after delivering two doses within an hour, thus preventing overuse and misuse. The number of remaining doses is displayed to assist the patient with adherence and has a child-resistant cap for added safety.”
The device has been validated for Human Factors in accordance with IEC 62336—it is user-friendly for senior patients, provides adequate finger-space, a color-distinguished nozzle, and an easy to hold overall grip.
Optimized Implant Packaging from MULTIVAC Export AG; Stryker GmbH (Switzerland)
 World Packaging Organisation
World Packaging Organisation
This duo delivered an optimized implant package that allows medical professionals to handle products quickly and hygienically, meeting international standards that require these devices to be delivered to the point of use in clean and functional condition. “Usability requirements also demand safe and effective handling, while customers expect rapid unpacking. The packaging not only enhances hospital operations but also delivers substantial production benefits. Automated production increases efficiency, achieving up to fivefold speed compared to manual methods. Thermoforming enables convenient product placement, and automated labeling and content checks further streamline the process,” says the entry.
The system is also a fit for items previously packaged in rigid film trays, which could result in reduced waste while accommodating automated thermoforming vs. semi-auto bag and tray packaging.
Drug Package lnsert lntegrated Paper Container of Liquid Bandage from Kobayashi Pharmaceutical Co., Ltd. (Japan)
 World Packaging Organisation
World Packaging Organisation
“To address the issue of product misuse without reading a package insert, we have provided explanations on the inner surface of the package. The packaging is designed to be opened from the bottom of the case, making the usage instructions easily visible to users,” says Kobayashi. “Additionally, the removal of the insert has resulted in cost savings and reduced CO2 emissions. This package design with a header is commonly used in over-the-counter medical products, making it applicable to a variety of pharmaceutical products as well.”
PharmaGuard from SÜEDPACK Verpackungen GmbH & Co. KG (Germany)
 World Packaging Organisation
World Packaging Organisation
As companies in the pharmaceutical industry strive for more sustainable practices, the PharmaGuard blister concept is a recycle-ready, PP-based monomaterial package that offers a high level of transparency. “The bottom and lidding films are produced applying a unique coextrusion process. Both film structures are based on PP and are perfectly aligned to each other, particularly in terms of their sealing performance. This guarantees not only the highest packaging quality, but also maximum safety in a highly efficient process. Depending on the product to be packaged, the PharmaGuard films can be equipped with different barrier and mechanical properties for optimum product protection and best possible stability,” says the company. A Sphera lifecycle analysis found the concept had up to a 47% lower carbon footprint and reduced energy and water consumption compared to traditional PVC/PVDC 250/40 Alu 20 blisters.
ClearX®-High Performance Syringe System from TAISEI KAKO CO.,LTD. (Japan)
 World Packaging Organisation
World Packaging Organisation
Prefilled syringe demand continues to grow. However, biopharmaceuticals come with risks of protein aggregation induced by silicone oil, which is used to ensure smooth gliding of the plunger inside the barrel. Taisei reports, “Our ClearX for prefilled syringe can realize silicon-free dosage, and can reduce this risk. Biopharmaceuticals also have a risk of adsorption. This adsorption could reduce pharmacological active ingredients. However, it can be minimized by choosing the syringe and piston made of low adsorptive materials, like our ClearX. Furthermore, ClearX is ideal for tube stoppering in terms of vacuuming air in the syringe when filling drug.”
Child-resistant (CR) packaging from Jaakkoo-Taara Oy (Finland)
 World Packaging Organisation
World Packaging Organisation
The company reports that there is industry need for low-cost CR packaging from a single carton blank that is both easy to open for adults while maintaining CR properties for children, even after opening/closing several times.
The winner is a CR package comprising an outer and inner casing, in which the inner can slide out with the push of a button after the user breaks the initial seal. The package can be produced in automated assembly machines with low costs and consistent quality.
Cerviprime Gel – Sterile packaging system for woman’s personal hygiene from Zydus Lifesciences Limited (India)
 World Packaging Organisation
World Packaging Organisation
The system was designed to meet requirements such as product protection for up to two years’ shelf life, process support, smooth machinability, and simplified user experience. “Design, shape, calculated length and meticulous mold validation procedure adopted to produce smooth, flange-free final articles ensures pain free usage of product at intended area of human body,” says Zydus. “Final packaging system tested extensively to check leakage, transport worthiness, extractability and leachability and found suitable for intended use. It is very difficult to maintain consistent quality and sterility up to 2 years for any packaging system which is just assembled and not tightly/hermetically sealed however this device ensures consistent quality when tested extensively at regular intervals up to 2 years.”
Check the complete list of winners, including a medical sample bag in the Personal Care category, at WorldStar's winner page here.






















