Two packages, both developed to aid patients with metastatic breast cancer, tied for the Compliance Package of the Year award in the Healthcare Compliance Packaging Council (HCPC)’s annual competition.
Novartis Kisqali Dosepak and Lilly Verzenio Dosepak earned HCPC’s top honors as the most innovative pharmaceutical packages designed to improve patient adherence for the preceding market year, 2017.
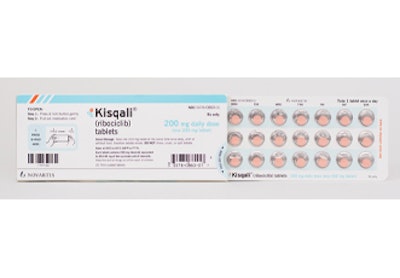
Novartis Kisqali Dosepak
Novartis Kisqali (ribociclib) Dosepak from WestRock-MPS is used for a solid-dose prescription medicine that works in combination with an aromatase inhibitor to treat pre/perimenopausal or postmenopausal women with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative breast cancer that has spread to other parts of the body (metastatic).
Kisqali requires three pills a day to be taken at the same time, each day for three weeks. Patient adherence to this regimen is extremely important. This award-winning medication package promotes patient adherence by implementing a custom design that integrates the following:
• A calendared blister clearly labeled by day on each individual pill cavity, highlighting the one-week dosing regimen (three one-week packages make up a monthly supply of Kisqali). Visual cues prompt patients to take and track medication for a three-week-on, one-week-off dosing regimen.
• A heat-sealed inner card, which allows for multi-color graphics for an easy-to-view calendar.
• Branding for Novartis/Kisqali on the outer package’s readable, flat space
One judge reviewing the package remarked, “The color-coordinated package provides graphic appeal and simple enough instructions. [A] pull-out tablet card and separate prescribing information/medication guide provide an appealing user experience, with easy-to-read type.”
Lilly Verzenio Dosepak
Lilly’s Verzenio (abemaciclib) is another solid-dose drug approved for hormone receptor-positive, HER2-negative metastatic breast cancer. It requires the patient to take both a morning and evening dose. Its packaging promotes patient medication adherence using a custom design that integrates the following:
• A calendared blister clearly labeled by day on each individual pill cavity, highlighting the one-week dosing regimen (four one-week packages make up a monthly supply of Verzenio). Visual cues prompt patients to take and track medication.
• A 5/6th panel that provides space for medication information, dosing instructions and how to navigate the calendar.
• Branding for Lilly/Verzenio on the outer package’s readable, flat space.
One of the judges reviewing the package stated, “Simplicity defines the Lilly Verzenio Dosepak. It opens without resistance. Instructions are remarkably clear. The package is easy to use, store and carry.”
“WestRock partnered with its customers to design packages to help metastatic breast cancer patients adhere to their medication,” said Dave Dwyer, VP, Global Marketing—Adherence, WestRock. “Packaging is the one tool that stays with the patient throughout the medication journey, making it uniquely effective in providing regimen support.”
The HCPC Compliance Package of the Year competition has been held annually since 1995. The not-for-profit trade association will present the awards for 2017 and the upcoming 2018 Compliance Package of the Year awards at their March 2019 HCPC members meeting when the organization will host the IoPP Drug and Pharmaceutical Packaging Committee in Charleston, SC.
Check out Part II of the story here.