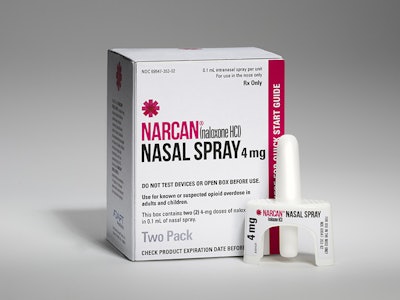
Last November, the U.S. FDA approved Radnor, PA-based Adapt Pharma’s NARCAN® (naloxone hydrochloride) nasal spray for the emergency treatment of known or suspected opioid overdose.
Aptar Pharma recently announced that it provided the delivery system and regulatory support for Narcan, which it referred to as “the first U.S. FDA-approved nasally administered, needle-free, ready-to use medication that can stop or reverse the effects of an opioid overdose.”
The liquid spray drug delivery technology platform provides dose accuracy and ease of administration. The single-shot unit-dose system incorporates a primeless feature and 360-degree orientation to offer the patient or caregiver ready-to-use convenience.
The two companies worked together to achieve the combination drug-device product compliance and regulatory approval.
“Opioid overdose is responsible for the deaths of thousands of Americans in communities throughout the country, leaving a trail of devastation for friends and families,” said Seamus Mulligan, Chairman and CEO of Adapt Pharma. “NARCAN Nasal Spray a ready-to-use, needle-free device, delivers a 4 mg dose of naloxone in a single 0.1 ml nasal spray. This new device makes naloxone readily available for emergency use by a friend, family member or caregiver, as well as offering an alternative treatment option for first responders and healthcare providers. It gives new hope to those concerned about the potentially fatal effects of opioid overdose.”
Narcan® contains naloxone which is an opioid antagonist. It is marketed in the U.S. for emergency treatment of known or suspected opioid overdose. “The U.S is facing an unprecedented opioid overdose epidemic. By making Narcan Nasal Spray available, we hope to increase everyone’s access to emergency treatments across the U.S.,” said Seamus Mulligan, Chairman and CEO of Adapt Pharma.
Aptar Pharma says, “Estimated at over $2 billion in 2015, the world market for intra-nasal spray systemic drug delivery has experienced strong growth in the last few years. The list of therapies successfully treated via the nasal route continues to grow from the traditional areas of migraine, osteoporosis and endometriosis, to now include: post-operative pain (ketorolac), cancer pain (fentanyl), prostate cancer, urinary incontinence, anemia and vaccinations.”