This content was written and submitted by the supplier. It has only been modified to comply with this publication’s space and style.
BD (Becton, Dickinson and Company), a global medical technology company, completed a preliminary study investigating the impact of deep cold storage (-20°C and -40°C) on glass PFS.
While traditional vaccine formulations are commonly stored up to about 2°C-8°C, new vaccine formulations – including those for mRNA – require much colder storage for over a long period of time to ensure drug-product shelf-life and potency [1][2]. Such low temperatures can induce changes in container dimensions, phase transition - liquid to solid and vice versa - and thermal shock. This could compromise the functional performance of the delivery system and Container Closure Integrity (CCI). BD began its investigation on the impact of deep cold storage on glass PFS as soon as information regarding the deep cold storage requirement for mRNA COVID-19 vaccines became available in 2020.
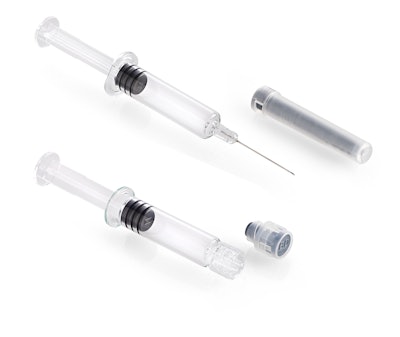
BD researchers conducted a comprehensive analysis of approximately 2,000 PFS after deep cold storage at -20°C and -40°C. Tests were conducted on several combinations of glass barrel coatings, formats - from 0.5 to 3 mL , different tip and flange designs, and multiple elastomeric closures with various state-of-the-art methods, including Lighthouse Laser Headspace Analysis [3], at these temperatures.
The study showed key product functions – PFS system level – were unaltered when stored at -20°C and -40°C. In addition, the count of subvisible particles and container closure integrity (CCI) remained unchanged when compared to those stored at room temperature.
“PFS are a known factor in addressing the complexity and costs of vaccine administration. They have been shown to significantly reduce time and labor associated with drug preparation compared to vial formats [4], and they support dose sparing [5]” said Bruno Baney, vice president of R&D for pharmaceutical systems at BD. “This study confirms the promising opportunity we have to apply our leading scientific capabilities to develop innovative delivery solutions for our customers now and in the future. We’re encouraged by these results and look forward to partnering with pharmaceutical companies to further advance PFS in deep cold conditions.”
Following its completion of this study, BD anticipates that BD glass barrel PFS systems should be suitable for use when storage temperatures of -20°C and -40°C are required.
Comprehensive study results were presented at the 2021 Universe of Pre-Filled Syringes and Injection Devices conference on October 6th.
Detailed study results are available to BD customers/partners upon request.
BD is also enhancing its manufacturing capacity and technology for PFS and advanced drug delivery systems with previously announced plans to invest $1.2 billion over four years.