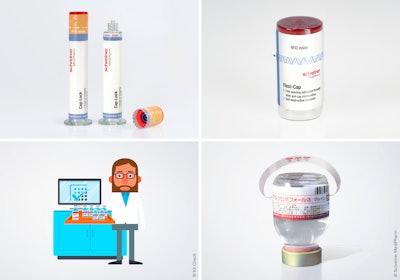
Schreiner MediPharm, a Germany-based provider of innovative functional label systems for the pharmaceutical industry, has collaborated with RFID tracking systems provider Kit Check to introduce RFID-Labels that are designed to allow hospital pharmacies to track and manage medication inventory and usage via intelligent software.
Error-free medication management in hospitals can save lives. Medicines equipped with RFID-Labels are made to be tracked from the hospital pharmacy to bedside dispensing meant to also have availability ensured—but only if the RFID-Label and tracking system are sufficiently synced. The new partnership between pharma label specialist Schreiner MediPharm and Kit Check, with its RFID tracking software and hardware, expends efforts to answer this need.
For example, using the joint system, a large number of syringes and vials equipped with Schreiner MediPharm’s RFID-Labels can automatically be scanned by a Kit Check scanning station to identify batch numbers and expiration dates. Subsequently, an inventory report should be generated to show any missing or expiring medications. This not only saves hospital staff significant time but, above all, reduces the risk of medication errors and contributes to patient safety, says Schreiner MediPharm.
Compatible and high-performing RFID-Labels are a prerequisite for Kit Check’s viable RFID systems. To ensure compatibility and performance, the company reviewed the label manufacturer’s technical capabilities and compliance with Kit Check's standards around certified inlays, EPC (Electronic Product Code) standards, serialization activities, and quality processes. If found to match Kit Check’s requirements, the supplier is awarded the “Works with Kit Check” trust mark as a certified manufacturer.
Stefan Wiedemann, senior director Strategic Marketing and Business Development at Schreiner MediPharm, explains the reasons for the cooperative partnership: “Kit Check’s innovative tracking software and hardware based on RFID is an ideal fit for our portfolio of smart label-integrated RFID systems. The design of our customized, highly robust labels ensures reliable readability via the Kit Check systems, thus enabling a safe and efficient supply chain in hospitals.”
Tim Kress-Spatz, co-founder of Kit Check, also welcomes the cooperation: “We know that Schreiner MediPharm has a track record of bringing unique innovations to market in the pharmaceutical space, including functional labels with integrated hangers, tamper-evident features and tactile security elements. We are excited to see the joint innovations that we are going to launch through this partnership.”






















