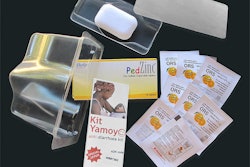
Fresh off earning honors as DuPont’s premier Diamond and Special 25th Anniversary Food Security award-winner, the AidPod/Kit Yamoyo pack continues its one-year trial run in Africa for ColaLife.
ColaLife is an independent nonprofit organization that leverages the Coca-Cola Co. (TCCC) distribution network to transport the AidPod packages containing medicine and information to the needy in remote areas.
AidPod is the name of a registered distribution packaging system whose design inspiration was to utilize the space surrounding bottles in a crate of Coca-Cola during distribution. The first application of this technology is for the wedge-shaped anti-diarrhea Kit Yamoyo (“kit of life”) package that nests or “piggybacks” between bottlenecks within the crates to minimize transportation cube. The ColaLife Operational Trial Zambia (COTZ) concludes in September.
In an effort to learn more and gain an update about the trial findings, Healthcare Packaging conducted the following question-and-answer communication with Rohit Ramchandani, B.Sc., MPH, Doctor of Public Health (DrPH) Candidate Department of International Health, Johns Hopkins Bloomberg School of Public Health, and Public Health Advisor & Principal Investigator, ColaLife.
Healthcare Packaging (HCP): What is the latest news with regard to this trial for ColaLife?
Ramchandani: We are currently in the process of finalizing our mid-line findings. After finishing-up with fieldwork in April, we have been busy cleaning and analyzing the data from our approximately 2,400 household surveys and 180 retailer surveys, from both our intervention districts and our comparison districts.
Early trends are positive, and give us confidence that we are moving in the right direction. For example, with regards to coverage, at midline we found that 31% of households had used Kit Yamoyo to treat a child with diarrhea. Perhaps even more impressive, is the fact that children who had never used zinc before are now doing so and benefiting from the globally recommended treatment regimen.
Prior to the trial, all the ORS (oral rehydration salts) was accessed in the public sector. At midline, we found that of those children in the intervention districts (where the Kit Yamoyo was available) with diarrhea in the two weeks preceding the survey, 43% used the Kit Yamoyo and sourced their ORS from the private sector. We suspect that this shift may go a long way in reducing the burden on an already strained public health system by freeing up capacity for higher priority issues that cannot be simply treated at home. We are in the process of gathering the necessary evidence to try and demonstrate this.
We have seen important improvements in terms of proper utilization of ORS, due in part to the Kit Yamoyo container serving as a measuring, mixing, and drinking vessel. We believe that if people are given the tools to make the best decisions for their health, we can improve outcomes. We are also seeing greater satisfaction scores in Kit Yamoyo users as compared to non-Kit Yamoyo users with regard to the taste and acceptability of the ORS by children. The ORS found in the Kit Yamoyo has an orange color and flavor, while the typical ORS does not.
The findings have also highlighted areas where we may be able to continue enhancing and strengthening the model and the Kit itself, to improve access and maximize benefits for mothers and children living in rural Zambia. For example, our midline findings showed that 80% of caregivers used four sachets of ORS or fewer. When designing the Kit, we erred on the side of caution, and included eight sachets to ensure children would get what they needed. Our findings demonstrate that in the majority of cases, this was more than enough to treat the child's episode of diarrhea.
This means that there will be ORS sachets left over from the first use of the kit and it is therefore likely that when diarrhea strikes again the child will be given the remaining ORS. In this case, the child would not gain the maximum treatment benefit due to the lack of zinc (which would have been used up if taken correctly during the initial episode). So, one thing we are doing, is reviewing the kit contents. This will help reduce costs and encourage adherence to the combination therapy of ORS and Zinc.
HCP: Please explain what is meant by mid-line findings?
Ramchandani: In order to test the impact of our model, we are conducting a rigorous scientific evaluation of the current one-year pilot. The findings from this evaluation will make up part of my dissertation at John Hopkins Bloomberg School of Public Health.
The design of the evaluation is called a pretest posttest design with comparators. This means that we gather data on a number of key performance indicators before and after the pilot in order to see what change took place due to our intervention.
For example, were we able to increase access to these essential medicines? Were we able to improve proper utilization of the medicines? Is it now easier for mothers to access these medicines, etc?
The study is being conducted across four rural districts of Zambia. Two of the districts serve as intervention districts where we are implementing the ColaLife model, and two others are serving as comparisons where the intervention is not taking place (these serve as counterfactuals—what would have happened in the absence of the intervention?).
HCP: How successful do you feel it's been?
Ramchandani: So far so good. While we know there is a need, it is good to see that we have also been able to start creating a demand for Kit Yamoyo and the medicines it contains. The communities we are working in are very happy with the product and we are seeing positive trends in a number of our key performance indicators. We are also aware of the challenges that exist in changing behaviors and improving health systems. At the end of the day, we have to let the evidence speak for itself, and we still have a number of months left in the trial, but as from the start, we are continually working to help facilitate the best possible options for the communities we are working with.
HCP: Please elaborate on what areas you might strengthen with the kit and kit packaging for future use?
Ramchandani: An important part of our work is to continually build on the findings from our research and what we are learning along the way.
Given all the recognition our work has received recently, which we are grateful for, it would be easy to rest on our laurels, however, one of the main reasons for conducting the pilot is to learn as much as we can so that we continue to improve the model so that the benefits will reach as many mothers and children as possible.
Part of this is to continue to refine and improve the kit while maintaining all of its functionality—Kit Yamoyo continues to serve as a measuring, mixing, drinking, and reusable storage container—and bringing down the costs. We are working closely with pi global to explore ways in which to do this.
HCP: What changes might be coming in the future and is there another trial or application in the works?
Ramchandani: As per some of the findings above, we are continually trying to incorporate learnings and refine the ColaLife model to enhance its sustainability and improve health outcomes for children.
This one-year trial will conclude in September. Thus far things seem to be moving in the right direction, but we must let the data speak for itself. Founders Simon and Jane Berry, and all of our partners are excited for what the future holds.
There is definitely scope for scale-up in Zambia, and there is interest from other countries to adapt the model to their own context and continue learning. We'll continue to build on the evidence and lessons learned and see where it takes us.