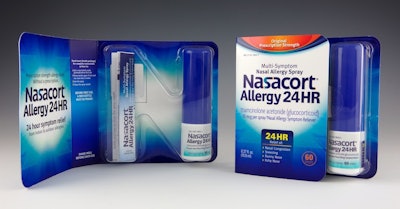
If a museum of contemporary over-the-counter pharmaceutical package design were created, it would likely display the Nasacort Allergy 24HR pack from Sanofi Consumer Healthcare Division Chattem Inc., Chattanooga, TN.
Nasacort Allergy 24HR spray provides full prescription-strength 24-hour scent-free and alcohol-free relief for nasal congestion, sneezing, runny nose, and itchy nose.
Nancy Limback, Sanofi Manager, Package Development and Innovation, says Nasacort started out as a prescription-only product, but was switched to over-the-counter sales, where it’s now sold in mass merchandising drug stores, retailers such as Wal-Mart, grocery stores, and many other outlets.
Nasacort became available in the first quarter of 2014 in 12- and 60-count spray varieties. Both use the same primary package, featuring an attractive wraparound paperboard pack that includes a thermoformed letter “N” for Nasacort, which holds the bottled product.
Limback says that in July 2014, a two-count 120-spray version was also introduced in the same thermoformed package. There’s also a three-count club package. The average retail price is $13.99 for the 60-metered dose spray and $18.99 for the 120-metered dose spray.
The thermoformed letter approach mimics the company’s previous package design strategy it used to roll out its Allegra allergy medication trial pack about two years ago. In that instance, the company used a thermoformed letter “A” to help the product stand out on shelf (see story here).
‘First-in-class’ product/package
Limback says the OTC versions represent “totally different packaging appearance compared with the Rx version, which was packaged in a carton printed primarily in white, white with a couple of different colors of print on it. “There was a total revamp for the retail market,” she notes.
Nasacort represents what Sanofi refers to as a “first-in-class product” launch for what is considered an intranasal corticosteroid drug.
“We really wanted to grab the consumer's attention,” says Limback. “It had to stand out. There were cartons and blisters for allergy products on the market, but nothing that had this kind of combination to it. We believe it’s the first in this category of retail nasal sprays in this type of package. Doing that was helpful in making it different on the shelf.”
Besides providing shelf presence to spark sales, the clear thermoform allows consumers to see the bottle of product inside, which isn’t always the case with competitive OTC allergy relief products. “The new pack has helped us increase sales and build our consumer base,” she says.
Secondary packaging considerations
The pack opens much like a book, with a cover and a perfect-bound spine. The front panel provides basic product name and purpose. The back panel and spine are printed with drug facts. Once the pack is opened, the left panel offers area for additional graphics or a coupon for a future purchase. The right panel contains the “N”-shaped blister. In the left vertical portion of the N is a folded insert with usage instructions. The right portion of the vertical N holds the labeled spray container.
“We thought it was very important to provide sufficient graphic area that not only carried regulatory copy but adequate information on its usage,” explains Limback.
The booklet card is printed on both sides, and holds the thermoform and a patient information leaflet inside the thermoform.
Although the OTC pack is noticeably different from its Rx predecessor, Sanofi did want to incorporate some design elements and match the barrier properties of the prescription carton.
“We utilized low-migration ink and coatings on the card application,” says Limback. “We wanted to avoid direct ink contact or coating contact with the bottle held within the thermoform.
“Also, we wanted to provide circulation within that secondary package to approximate the atmospheric exchange in the Rx carton so ventilation ports were added to the booklet card. You see those little squares in the back of the card and we also added ventilation panels in the seal areas of the thermoform itself. Then we had to also carefully design an area to add the electronics security tags so they weren't in contact with the bottle.”
The security tag adheres to the card, beneath the patient insert.
The packaging process
Limback says that Sanofi worked with MPS, the leaflet supplier, to evaluate different paper and fold combinations so that the company could achieve functionality without requiring adhesives.
The product is manufactured and filled into high-density polyethylene bottles from Nolato Jaycare with extended-content labels from Clondalkin Group in a Sanofi U.K. facility, then sent to the U.S. for secondary packaging. An updated opaque blue polypropylene pump overcap from Nolato Jaycare was added for a more OTC appearance. The pump assembly is made by Aptar.
Rohrer Corp. provides the thermoform and booklet card. CCL helped develop the booklet seal to be reclosable on the shelf.
“We were trying to marry two dissimilar surfaces, the thermoform and the card, and we tested a lot of different adhesives and labels,” explains Limback, “until we ended up with the patterned adhesive pressure-sensitive label you see.”
Contract packaging
Although Sanofi Chattem has its own packaging lines, contract packagers provide some capabilities the manufacturer does not have. For Nasacort packaging, the filled and labeled bottles are shipped to contract packager Reed-Lane for secondary packaging.
Reed-Lane, says Limback, uses a semi-automatic line that picks up blisters, places them on the thermoform, fills the bottle of product, folds the card over, the applies a seal to shut the package. Then they are collated into trays, shrink wrapped for a bundle and case-packed. A reclosable pressure-sensitive label holds the package closed. Specific machinery details and processes are considered proprietary, although Limback notes that Reed-Lane did require some new automatic and semi-automatic equipment for secondary packaging of Nasacort.
Special challenges
Limback says the thermoformed “N” posed special challenges. “One of the requirements that we looked at was that this package had to stand on its own, and it also had to function within pusher systems on retail shelves,” she explains. “We also wanted it to be memorable to the consumer. We had a whole list of design inputs that we tried to meet. So the way we designed the ‘N’ specifically to keep the round bottle from rotating since we couldn't use adhesive to hold the bottle in place. The thermoform and overall finished package is consistent across the face of the package in depth to operate efficiently in retailer shelf pusher systems. It also contains ventilation channels. The on-shelf product visibility provided by the ‘N’ thermoform also addresses the California slack fill requirements. There’s a substantial amount of functionality built into this package.”
The 15-mil PET thermoform uses 40% recycled content, an important consideration when selecting materials, says Limback. “It is definitely one of the considerations that we had on our design input. With this package we tried to utilize as little paperboard as possible. It is an 18-point board made from SFI-certified paperboard approved by the Sustainable Forest Initiative. It’s printed offset in seven colors. The thermoform is also recyclable, so yes, we definitely keep that in mind as we developed the package.” Rohrer Corporation provides both the thermoform and the paperboard components.
In all, Sanofi employs graphics punch, package shape, and sustainable materials to reinforce the Nasacort brand image. Limback suggests, “Maybe a consumer can't remember the exact product name but hopefully they will remember the package.”
Limback praises the work of The Goldstein Group for “doing a bang-up job on the graphics and working with us to coordinate with the package development.
“We leveraged a cross-functional, cross-divisional global packaging team that worked on developing this,” she says. “It certainly wasn't done by package development alone sitting in a vacuum. Marketing was extremely supportive of our innovation efforts, our R&D group over in the U.K. worked right alongside with us on the use of low migration inks and use of the ventilation ports, and our key external partners had a significant part in making the package a success,” she says.
Limback says that early input from retail channels and consumers has been “very, very positive. We did talk with key retailers and channel marketing as we developed this to satisfy all their requirements. I believe the retailers are very thrilled with it.”
On top of that, the package has already earned industry recognition as a finalist in the Sanofi Global Innovation Contest as well as won an AmeriStar Award.
“For many years our team walked through PACK EXPO and we saw winning AmeriStar package exhibits,” she says. “And now, to have one of our products actually appear in that exhibit made my bucket list.”
In addition, the Nasacort packaging was awarded a 2015 World Star Award.
Watch a video on the product here.
Nancy Limback will be speaking on “Packaging Innovation—Method or Magic?” on June 10 at EastPack.