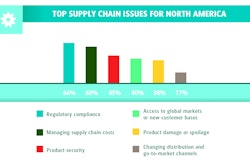
Due to extensive delays enforcing the 1988 Prescription Drug Marketing Act (PDMA) and a lack of specific traceability requirements in the PDMA, the State of California presented the industry with the concept of electronic pedigree and chain-of-ownership tracking. In 2004, the California Board of Pharmacy passed legislation aimed at more aggressive consumer protection due to the growth of counterfeit and diverted drugs.
The law required all pharmaceutical supply chain partners, from manufacturer through pharmacy, to pass along an electronic pedigree detailing the movement and transfer of ownership of a pharmaceutical product at the smallest salable item level.
With California comprising what some have said is the ninth largest economy in the world, industry had to step up and take notice.
This mandate was initially supposed to take effect January 1, 2007 and was later pushed out to 2009, then again to 2011. In 2008, after continued meetings with industry stakeholders, the California Legislature and the California Board of Pharmacy agreed to again push out the effective dates to 2015 through 2017.
These initial delays, and target date “creep,” have unfortunately set the scene for industry to hold off on implementing, in the hopes that further delays (or a federal override) may occur.
If your company has begun activities intended to ensure compliance with the California mandate, is that work all for nothing, now that it looks like the DQSA will pass and supersede it?
That depends.
For serialization compliance, there are differences. California only mandates that the NDC and serial number be on the product in a machine-readable code. H.R. 3204 legislation would mandate that, plus the lot number and expiration date in the same code. Numbers aren’t available, but from anecdotal evidence it appears that many companies were already planning to include the lot and expiration date in their barcodes for California even though they are not required. For those companies, DQSA would not add any extra burden for serialization compliance.
But companies that were planning to do the minimum for California will need to do some more work. For those that were planning a slap-and-ship approach to serialization for California, lots of additional work will probably be required because of the lot and expiration date requirement of DQSA. On the other hand, systems to produce aggregation data to support the use of inference on shipment and receipt appears to be put off for up to 10 years.
Not only that, but the lot-based nature of the DQSA transaction information, transaction history, and transaction statement paper or electronic documents will cause some companies to need significant adjustments to their e-pedigree solutions, which were predominantly focused on the package-level serialization required in California. In fact, will companies even need the same level of sophisticated systems to implement the exchange of these new lot-based documents? We may not know until the FDA publishes its standard—one year after the law is enacted.
Yes, it all started with California, but eventually companies will probably ask, does it all end with DQSA?
Liked this article? Download the entire playbook here.