Wright Medical Group, Inc., is an Arlington, TX-based global orthopedic medical device company that designs, manufactures, and markets reconstructive joint devices and biologics.
Recently, the company sought protective, safe packaging that could universally fit a variety of different sized and shaped extremity implant screws, many of which had sharp pitches or tips, for the foot and ankle region.
In an effort to design packaging and create efficiencies of scale, Wright worked with Placon, a designer and thermoformer of custom plastic packaging, and its Barger medical division.
Placon and Barger developed a PETG clamshell package for some 350 SKUs that was recently recognized with an IoPP Ameristar Award in the Medical Device category.
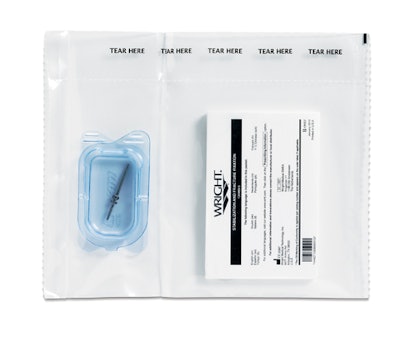
Designed with beauty and simplicity in mind, the small, distinctive rigid thermoformed clamshell meets Wright’s requirements for form, fit, and function. When closed, it measures 2½ in. long x 1½ in. wide x 0.340 in. tall.
Designed for use in both sterile and non-sterile presentations, the PETG thermoform utilizes a single, consistent footprint for easy product validation. The thermoform has no sharp edges that come into contact with the primary packaging material, ensuring no punctures or pinholes to the package while in transit or during storage and handling.
The packaging makes it easier for operating room (OR) staff to easily find the part within the sterile packaging configuration or non-sterile plastic bag without having to “dig” in search of the product. OR staff is able to open the inner sterile barrier with enclosed thermoform for aseptic presentation directly onto the tray stand in the sterile field. The pack also helps eliminate the need to dig for the screw or to turn the bag upside down and risk screws flying out and possibly rolling off the tray and onto the floor.
With a universal system developed, Wright has streamlined its entire packaging process. The end package has delivered benefits that include making handling easier, reducing inventory needs and validation costs, and providing a safe, reliable, and user-friendly protective package.
'Coining the hinge'
Sharp edges and snags are serious packaging problems, especially in the OR. Snags on a package can lead to punctures in sterile pouches, making medical devices non-sterile prior to being received in a sterile field. Sharp package edges, commonly associated with a package’s hinge, could also cause grievous rips in surgical gloves.
To help allay this issue, Barger employed a technique more often seen in the plastic injection molding industry. Instead of thermoforming the clamshell’s hinge in a “W” shape, whereby the hinge is cut on a plane, the hinge instead was “coined.”
With crushed cut hinges shaped in the form of a “W”, hard edges can be produced, leading to the possibility of potential snags in sterile bags or pouches. In addition, “W”-shaped hinges typically can’t close flat without some gaping around the perimeter. To avoid these common issues, a coined or “living” hinge was created in the mold with a mechanical anvil pincer to make the mechanical hinge lay flat, with the added capability of being able to be flexed hundreds of times without breaking.
The only potentially sharp area on the clamshell lies on the corners of the hinge. To assure that pouches are protected from this potential area of concern, two rounded “wings” were designed on either side of the hinge. This simple design addition eliminated the possibility that the pouch could come into contact with potential sharp hinge corners when the clamshell is closed.
Developing a streamlined protective packaging system that could house and protect a large number of orthopedic implant screws of varying sizes and configurations was a challenging task. In addition, it was critical that the packaging could be properly sterilized while maintaining the implants’ physical integrity through the filling, distribution, and handling processes.
With a large number of different size and shape components, Wright initially used a varied assortment of packaging materials to hold the components, from plastic bags to urethane/silicon-type end caps to foam piece cavities. However, due to the vast amount of sizes, an equally broad number of configurations needed to be acquired.
Redesigning for product form, fit, and function
Before the universal clamshell was designed, Wright relied on different packaging materials to house the various-sized parts, all with different success rates. Silicon/urethane-type end caps placed on the screws tips were abandoned due to the number of parts Wright needed to inventory. The company also found that it was difficult to fit each screw’s exact form with the end cap; some caps would fit too snugly while others, too loose.
Direct placement of the screws into a plastic bag was also tested, however, it was discovered that specific parts were experiencing varying levels of damage. In particular was Wright’s snap-off screw, a part that spins/drills into the bone. When the part is unable to be maneuvered in any further, the shaft is designed to snap off. However, when this part was stored directly in the plastic bag, and the shaft was prone to bend or even break off when a certain amount of direct pressure was applied to the part during shipping or storage.
Finding the right balance
Requirements that Wright specified from the onset of the design process included developing packaging that:
• Had high performance capabilities. The design needed to securely protect the implant screws while ensuring that the packaging’s sterility was upheld, with no abrading of parts, no pinholing, or film fracturing of pouches.
• Could handle a multitude of various sized and shaped implants.
• Could withstand ethylene oxide (EtO) and gamma sterilization.
• Would be user-friendly.
The thermoform design addressed a number of concerns including:
• Reduction of inventory costs. The universal packaging eliminated Wright’s need to carry multiple sizes of different materials such as silicon end caps or foam. The rigid PETG container has the flexibility to accommodate a wide assortment of different-sized screws into a simple package with a single footprint. The fewer packaging components equated to less part numbers to manage and track.
• Reduction in validation costs. Although the tooling costs were greater than the cost of standard tooling costs, Wright made up for the larger upfront expenditure through the ease of validation. Validation was only required for one package versus several packaging configurations. In addition, testing/QA procedures were consolidated due to the smaller number of packaging configurations.
• Reduction in future production costs. With one common footprint, Wright was able to utilize 20-up clamshell tooling which helped decrease costs in future production runs due to standardization of tooling.
• Reduction in labor costs. The manually filled packaging is easier to fill thanks to a one-size-fits-all thermoform and a reduction in packaging components.
• Reduction in product damage. Returns due to damage have been virtually eliminated due to the new packaging design
The clamshell has received positive feedback from not only the end-user/customer side but from the back-end or assembly side as well. Packaging line assemblers who manually fill the entire carton have noted its complete ease of assembly. Assemblers are able to drop in the part, close the thermoform, and place the thermoform inside a non-sterile packaging configuration or a sterile pouch-packaging configuration.
Sterile and non-sterile presentations
With the ability to work in both double-barrier sterile or non-sterile presentations, the thermoform is multifunctional in use. When utilized for non-sterile presentations, a double-compartment plastic bag is used with the IFU sealed in one compartment and the clamshell in the other. Non-sterile packaging is used in distributor instrument cases. For surgeries that may require multiple tool boxes, distributors place non-sterile products in instrument kits, which are then sterilized at the hospital to be autoclaved.
Initially designed for the IFU to be inserted alongside the clamshell in a single compartment, Wright moved to a double-compartment bag to ensure adequate separation of the IFU and component from each other so that the component would not stick in-between the IFU pages.
For double-barrier sterile uses, a screw is placed in the clamshell and sealed into a chevron plastic Tyvek® pouch that is subsequently sealed into a second chevron plastic Tyvek® pouch that is slid into a carton with the small IFU. The complete packages are either sterilized by gamma radiation or EtO gas. Channels/gates designed on the side of the thermoform allow EtO gas to flow through to the inside of the closed clamshell, sterilizing the screw and the entire package.