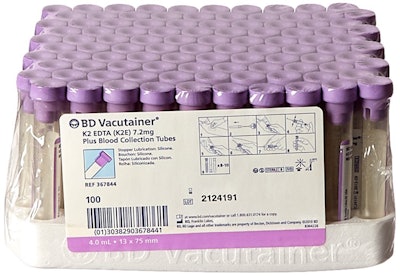
The whole point of a sealed blood collection tube is to protect the contents from contamination so that they may be used in diagnostic tests. However a recent FDA MedWatch article said Vacutainer blood collection tubes by Becton Dickinson are being recalled for doing the opposite. Apparently a chemical in the rubber tube stopper interferes with the accuracy of the Anodic Stripping Voltammetry testing methodology, which is used in Magellan Diagnostics’ LeadCare Testing Systems.
The chemical in question is thiuram, which can release gases containing sulfur that dissolve into blood samples and bind to the lead particles. This process makes it hard for the lead tests to detect the right amount of lead, yielding inaccurate results. BD is recalling all collection tubes with rubber stoppers in colors lavender, tan, pink, and green.





















