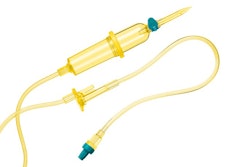
Baxter 50 mm 0.2 Micron Filter / Photo: Baxter
A notice issued by the FDA says Baxter Corp. is recalling all unexpired lots of their 50 mm 0.2 Micron Filter. The device is used in hospital pharmacies to filter bacteria when preparing water-based solutions used in IVs.
Baxter received reports about the presence of particulate matter and the potential for missing filter membranes within the device. The recall is identified as Class 1, which is the most serious type of recall that could lead to serious injuries or death.
Companies in this article