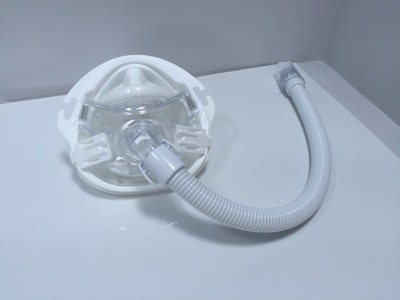
The package provides a positive experience for the patient. The Amara View CPAP is a minimal contact oral/nasal mask design. Its package, the Amara View Bubble, captures and contains the mask without contacting its delicate silicone membrane. A nose feature was molded into the package to provide patients and clinicians with a visible cue of how the mask functions.
These benefits helped the Amara View Bubble earn the coveted Best of Show award for Plastic Ingenuity in the Institute of Packaging Professionals (IoPP) 2016 AmeriStar Package Awards Competition.
A global company in the sleep and respiratory markets, Philips Respironics says Amara View’s “innovative design prevents red marks, discomfort or irritation on the nose bridge.” The company reports it “is the smallest and lightest of all the leading full face masks and is easy to use with one modular frame for all cushion sizes.”
Rob Helmke, Plastic Ingenuity’s Marketing Director, provides insights on the key aspects of the Amara View Bubble:
• Innovation. “One of the biggest challenges was containment of the mask to the package as there are no straight edges on the faceplate of the mask to snap onto. To minimize the use of packaging, a two-piece containment design was not an option. This [led] us into the claw design to snap onto the clip areas. In addition, Philips requested a design that was similar to a human face to visibly convey how the mask was to be used.”
• New design material/technique. “We had to float the part on a post feature in the middle of the package, while allowing the mask to be easily removed from the package. However, the post alone did not prevent the mask from rotating and popping off, which is why the design includes two extending claws to attach to the post features of the mask. These claws help secure the package to the mask, while [also] providing the patient with the awareness of how the headgear clips will attach.”
• Creativity. “In order to function, the claws must wrap around the posts on the mask. A regular fixed feature on the mold wouldn’t be able to accomplish this because of stripping issues and deformed parts. To address this, we used pneumatic flippers that retract those portions of the mold back into the base, so it can be pulled off the mold without damaging the parts.”
• New material applications. “We used 0.030-inch high-impact polystyrene material with an impact modifier. It is a very difficult draw with the post needed to suspend the mask. HIPS can also withstand the heat deflection of 160°F, which helped drive the material decision in addition to the impact strength properties.”
• Protection. “The mask packaging must protect the sealing membrane from possible compression setting due to extended storage times in warehouses and transit through the mail to patients. Within the overall package are large instruction booklets, which have sharp corners that could potentially puncture the membrane, and the final purpose is to reduce the amount of dust and debris that will stick to the membrane. Due to the tacky nature of the silicone, it attracts dust, fabric fibers, and other debris within the package.”
• Testing methods. “Masks are required to be tested in their ‘as shipped’ condition, which means in their final packaging, and within a fully loaded corrugated shipper box. Testing is performed according to the International Safe Transit Authority [ISTA] 1A vibration and drop tests. There is also a required environmental test to simulate the possible environmental conditions (extreme hot and cold, and humidity). These testing methods simulate the worst possible conditions the masks could see during transit/storage and are accepted by the FDA. At the completion of these tests, masks and packaging must not be damaged, deformed, or show any cosmetic issues before being considered acceptable.”
• Economics. “There are four different sizes of masks that utilize this single package, which minimizes SKUs on the customer’s end so they are able to maximize ordering volume and receive large-volume pricing efficiencies.
• Performance. “The parts are nested in a box then manually extracted and assembled. The mask self-locates on the center post of the package, then the claws are engaged to secure it in place. The headgear straps are tucked into the t-slots on the sides of the bubble, and then bundled on the back side. The attached hose wraps around the package for proper organization and to prevent contact to the front of the mask. Then the assembly is slid into a printed pouch, with printed instructions, and sealed.
• End-user benefits. “Due to the innovative design of the Amara View mask, which requires the mask to cradle under the nose rather than fit over the nose bridge, a nose feature was molded into the package that provides both patients and clinicians with a visible cue of how the mask should rest under the nose.”
• Environmental impact. “This is a package made from monolayer HIPS and can technically be recycled. There is just enough gauge to form a functional part, yet be rigid enough to support the mask. Recycled material includes 50 percent industrial-grade HIPS.
The 2016 IoPP competition judges evaluated and analyzed more than 80 packages from 14 categories in an in-person judging process in April 2016. Judging was based on package innovation, materials use, sustainability, protection, economics, performance, and marketing.