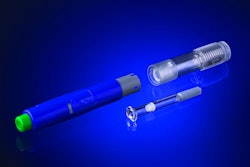
BD Medical, a segment of BD (Becton, Dickinson and Company) announced the commercial launch of a new passive needle guard technology, BD UltraSafe PLUS™, which has received 510(k) clearance as an anti-needlestick safety device.
The easy-to-use one-handed device is enhanced with ergonomic features designed to facilitate comfort and support for healthcare providers and patients. In addition, this safety device is designed to meet increasingly complex biotechnology drug requirements, including higher viscosity. The device allows patients the ability to manually control their injection.
The BD UltraSafe PLUS Passive Needle Guard provides includes a robust plunger rod to help support injection of viscous drugs, a larger drug inspection window to improve drug visibility, extended built-in finger flanges, and an enhanced plunger head for improved injection support and stability. These ergonomic improvements provide support for users, including patients with diminished manual dexterity.
BD recently conducted a clinical focus group with patients who had reduced dexterity and also suffered from rheumatoid arthritis and multiple sclerosis. Subjects reported that the BD UltraSafe PLUS Passive Needle Guard provided ease of use with 100% of injections executed successfully. The Passive Needle Guard is designed to enable easy integration into existing safety device assembly lines and is compatible with ISO standard prefilled syringes.
The BD UltraSafe PLUS Passive Needle Guard is designed to be used in conjunction with 1.0 mL-long prefilled syringes with staked needles, such as the BD Hypak™ or BD Neopak™ Glass Prefillable Syringe. The product was developed by Safety Syringes, Inc., an anti-needlestick device company acquired by BD in December 2012. The BD UltraSafe PLUS Passive Needle Guard received positive feedback when customers previewed the product at the October 2012 Parenteral Drug Assn. meeting. Orders have been received, and commercial shipments of BD UltraSafe PLUS Passive Needle Guard are expected to commence shortly.