Antares Vision, provider of serialization-based pharmaceutical track-and-trace products and inspection systems, introduces ATSFOUR, an enterprise-level serialization software solution that fights against counterfeiting in the pharmaceutical industry.
As new drug traceability regulations continue to be enforced in the U.S., Europe and other key global markets, the need for Level 4 software has become increasingly important. The new ATSFOUR integrates the Antares Tracking System architecture and is comprehensive and scalable to allow the widest connection capability available for any traceability framework. The software ecosystem manages the serialization data flow connecting production plants, CMOs, third-party logistics and authorities, and manages high volumes of data through an innovative data storage system.
The versatile, compatibility-minded ATSFOUR creates a unique point of connection enabling interface with healthcare agencies, CMOs and other brand owners operating with a variety of software and hardware solutions.
ATSFOUR stands out for offering two different installation options: not only on a cloud platform, but also on-premises in the local server infrastructure of the pharmaceutical company.
ATSFOUR can be configured on an entirely dedicated cloud space that is not shared with other companies. In view of the constant increase in the volume of data to be managed, ATSFOUR is designed with graph database technology, which allows for a faster search of information compared to traditional systems.
Many companies have already chosen to implement the ATSFOUR, including Recordati, Salf, Sit, Fine Foods, Marlex, biomo pharma, Renata, Faes, Saneca, Sovereign, Adhex, Sifi, Dms, Sirton, Promed and Falorni.
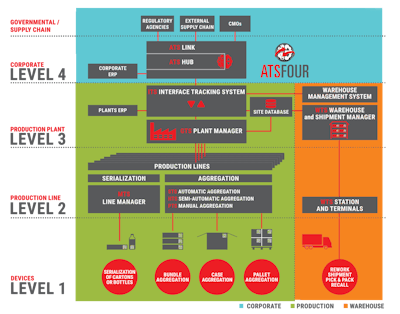
The comprehensive software solution allows pharmaceutical companies to manage information from the line level (Level 1) to the corporate/enterprise level (Level 4) and reporting to regulatory authorities (Level 5).