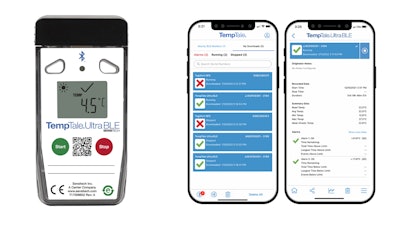
Sensitech, a leading provider of global supply chain visibility solutions, has launched the TempTale® Ultra BLE, the newest wireless addition to its line of digital temperature monitoring solutions.
The TempTale Ultra BLE is a validated Bluetooth temperature monitoring solution capable of reading data without opening cargo packages, providing delivery drivers and receivers proof of condition at destination. This new data logger offers an intuitive mobile app for touchless monitor control, clear evidence of alarms, and seamless synchronization to the system of record, speeding product release for receivers and quality managers.
With a thinner dimension, the TempTale Ultra BLE is ideal for smaller packages, such as clinical trial kits and direct-to-patient, pharmacy, and hospital shipments. Seamless, one-tap data synchronization to the system of record means couriers and receivers will avoid managing multiple cloud logins. Secure reports can easily be shared from the app.
“With the launch of TempTale Ultra BLE, we have retained our rigorous commitment to quality, while adding Bluetooth functionality as a time saver for shippers, couriers, and receivers,” says Henry Ames, general manager of life sciences, Sensitech. “The TempTale mobile app is an intuitive conduit for life sciences’ stakeholders to easily and quickly demonstrate proof of product condition at delivery, assuring patient safety and meeting regulatory compliance.”
The monitor provides -30 °C to 70 °C temperature range with six programmable alarms, following consistent TempTale quality and specifications. Each monitor is calibrated to 3-point National Institute of Standards and Technology traceable standards with an on-board certificate to meet data accuracy and regulatory requirements.
The solution includes Sensitech’s audit-ready 21 CFR Part 11- and Annex 11-compliant ColdStream® Select cloud software. To support all supply chain partners regardless of smartphone ownership, the TempTale Ultra BLE includes an LCD screen with familiar TempTale icons for immediate alarm status and a secondary USB for validated data retrieval.
“The ability to read through packaging when demonstrating proof of product condition at delivery dramatically reduces time, and it improves operating procedures while retaining packaging integrity at shipment waypoints,” adds Ames. “Not having to log in to multiple systems makes it easy to manage receipt of deliveries and return data to originating pharma companies, saving time, eliminating human error, and streamlining product disposition.”