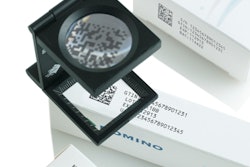
During this major European trade show event, Optel Vision will present its latest technology in Track and Trace - TrackSafe. TrackSafe serializes and labels individual cartons or bottles while ensuring that each serial number is valid, unique and readable. Once printed on the package in a human-readable format and encoded in a 2D and/or linear bar code, the serial number is again inspected with a camera for quality and validity control, and its status is registered and transferred to the data management repository. TrackSafe can also manage RFID tags encoding and reading as well as the whole serial numbers allocation and verification process and the transmission of the related data in an EPCIS compliant repository.
The Optel Vision solution presented at Achema will consist of a station with a four-camera mounting allowing for a 360-degree inspection for round bottles, plus a bottle bottom inspection camera. The configuration of this station allows for the association of the bottle label's serial number with a temporary code printed underneath the bottle. This way, the system can retrieve the serial numbers of the bottles during the bundle aggregation when their serial numbers are not visible.
Optel Vision is additionally introducing a new planning service to help companies to prepare for Track and Trace. This serialization strategy planning is very thorough. It includes gap analysis study, infrastructure planning, execution planning and review of all current requirements necessary to bring your corporation up to speed for serialization. This planning process will ensure that all your money used in Track and Trace is spent the correct way, no losses.
Optel Vision is also featuring monthly webinar session; to help companies to become familiar with the current and evolving serialization requirements and more importantly to review and understand real use cases of Track and Trace. If you are interested in joining our webinar, please send an email.
For more details on Optel Vision's Track and Trace solutions, visit here.
Companies in this press-release