As regional regulations pose increasingly unique supply chain challenges, more healthcare organizations are turning to import brokers to assist with shipping products globally. That’s one insight from a recent industry survey performed by Sonoco ThermoSafe, a global provider of temperature-controlled packaging, in partnership with Arena International Events Group and Berlinger Co. AG.
The survey, “Assessing the Current and Future State of Clinical Trial Supplies” was released this week, co-developed by an advisory panel of industry experts from clinical operations, clinical trial supply logistics and packaging, as well as bio-pharma manufacturing and supply chain organizations.
The survey was conducted between March and December 2016 and highlights forward-looking trends in areas that influence the clinical trials supply chain – including adoption of technology, scope and pace of supply chain innovation and logistics/distribution strategy. A total of 230 healthcare industry professionals participated in the survey, representing organizations from more than 20 countries and clinical supply operations across five continents.
According to Vishal Khushalani, Director of Global Marketing and Business Development at Sonoco ThermoSafe, the study revealed several key insights:
-
53% of respondents from the sponsor organizations reported they outsource more than 61% of their clinical trial supply budget.
-
Close and reliable partnerships between clinical and operational teams were cited as the most important factor in successful supply forecasting: relationships with CRO 87%, distributor/packaging supplier 85%, followed by the manufacturer 74%.
-
24% of respondents currently use a direct-to-patient (DTP) distribution model. One-third of those who don’t use DTP today plan to adopt this distribution model over the next 12-18 months, driven by the desire to optimize cost of recruitment, patient interactions and leverage efficiencies in data collection/visibility using technology and specialist service providers.
-
69% of respondents anticipate investment in mobile technology in the next two years. Lack of a user-friendly interface and data security concerns topped the list of factors limiting their company’s investment in mobile technology platforms.
-
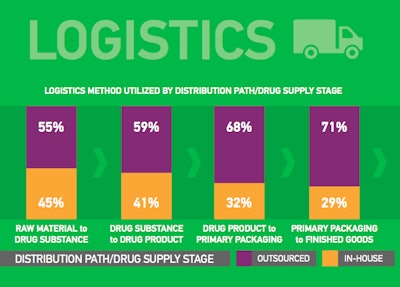
Respondents found logistics between depot and clinical sites most challenging in South America, Central America and Asia. Further, the survey indicates widespread use of clinical trial supply partners, with 86% of respondents using an import broker to overcome regional challenges such as compliance with import/export regulations for shipments of clinical trial supplies.
Russell Grissett, Division Vice President and General Manager of Sonoco ThermoSafe says, “The insights gained through this survey will hopefully help companies baseline their current strategies in clinical trial logistics, supply chain and technology and guide future investments.”
Detailed findings were presented at the 6th Annual Clinical Trial Supply New England conference in Boston, MA, on March 8, 2017, and will be noted at subsequent Arena International Group events across North America, Europe and Asia throughout 2017. Survey results in infographic form and a detailed report can be downloaded here.